High-Throughput Design of New Perovskite Oxide Antiferroelectric Materials
Introduction
Antiferroelectric (AFE) materials have recently been of a great interest, due to their unique applications in the energy storage devices and non-volatile random access memories. Most of the currently available AFE materials are lead based, which are environmentally harmful. So this unique application of AFE material in one hand and lack of lead-free AFE materials in the other hand, motivated us to study the cubic - AFE phase transition of NaNbO3, as a known AFE material, and the effect of doping on AFE to ferroelectric (FE) phase transition. This better understanding of NaNbO3 paves the way to design new lead-free AFE materials.
Methods
-
Cubic - AFE phase of NaNbO3
The density functional theory (DFT) calculations was performed to calculate the energy of the interpolated distorted structures. Later on, for Pseudo Jahn-Teller effect (PJTE), we did symmetry analysis and by unfolding the band structures we studied the adiabatic potential energy surface cross section for each mode.
-
0.9375 NaNbO3 - 0.0625 SrSnO3 solid solution
Many different Supercells with 80 atoms were generated for both FE and AFE phases to ensure having enough variant Sr-Sn distances. As the AFE-FE phase transition is Displacive, we also considered the corresponding structures.
Results
-
cubic - AFE phase of NaNbO3
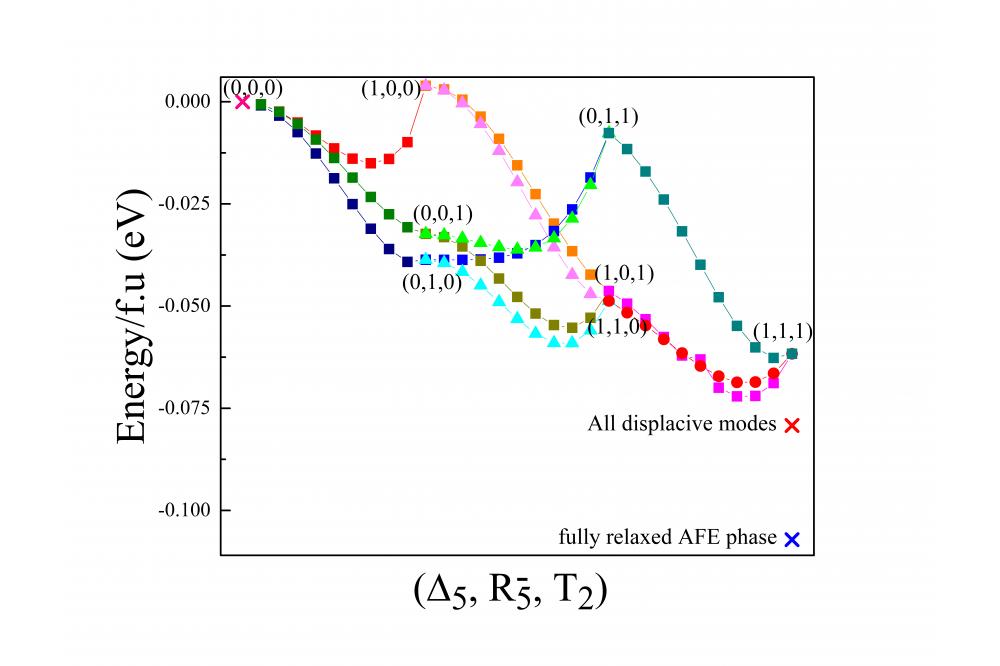
After relaxation and finding the distortion modes driving the phase transition through cubic paraelectric (PE) to Orthorhombic AFE phase, we found the potential energy surface (PES), as shown in Fig.1.a. Then by fitting the energies to the Hamiltonian model based on Landau theory, we concluded that R5- and T2 modes (the modes responsible for Oxygen rotations) gain more energy, while Δ5 mode (the mode responsible for AFE displacement of Nb and Na2 ions) does not decrease the energy, and it individually increases the energy. Biquadratic couplings showed that there is a strong repulsion between R5- and T2 modes if they coexist without presence of Δ5 mode. The gain of energy by the trilinear term denoted the crucial role of this term, as without such a coupling the AFE phase could not be stabilized.
Then we went one step further, and found the electronic force driving this phase transition by PJTE. The results showed that coupling of T2u ground state and T2g excited state, creating Δ5 mode, is the electronic source of instability. This shows the overlap of O-2p and Nb-4d orbitals which by increasing the covalency between Nb and O ions, creates Nb displacement as the distortion causing the instiability in the high-symmetry cubic phase.
- 0.9375 NaNbO3 - 0.0625 SrSnO3 solid solution
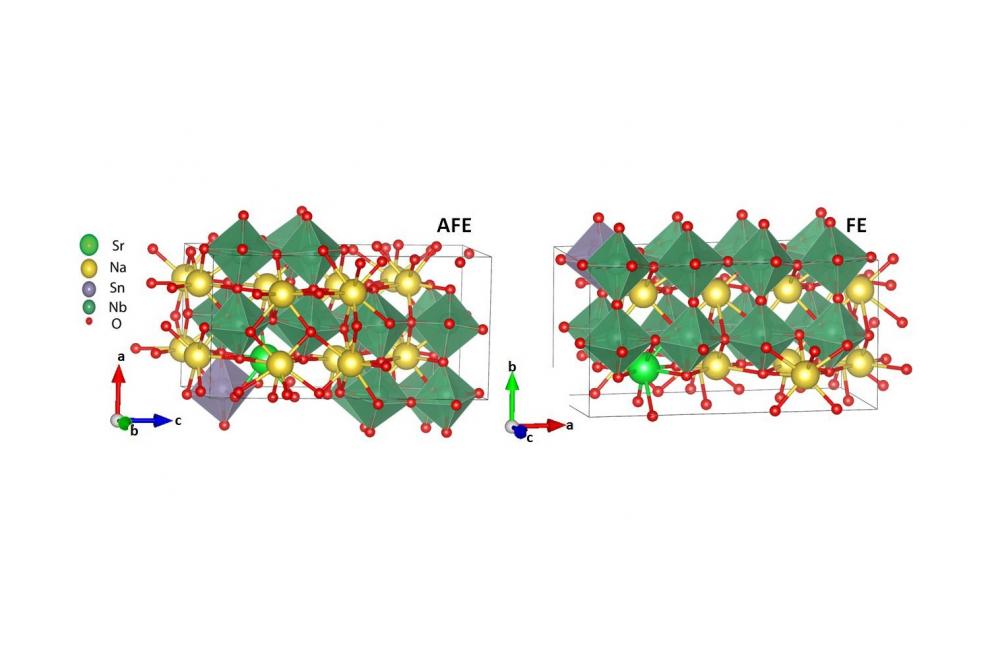
In the pristine NaNbO3 the energy difference between the AFE and FE phases is 2.3 meV/f.u., whereby the FE phase is more stable. This energy difference is further reduced upon Sr2+ and Sn4+ substitution and reaches a value of 1.4 meV/f.u. Interestingly, after this substitution, the AFE phase is more stable. Comparing the energy difference between the corresponding structures, also showed the more stability of the AFE phase over FE phase, by doping, in a good agreement with experiment. Two of the corresponded structures are shown as an instance in Fig.2.