London Dispersion Forces in Structure and Reactivity
Introduction
London dispersion forces help explaining unexpected attractive interactions of organic material. During the last century, London dispersion forces slowly slid more into focus of organic as well as inorganic chemists whereas their omnipresence in chemistry became more visible with every article published. In order to study those fundamental interactions a finely balanced system was designed to quantify London dispersion forces in molecular systems.
Methods
Design of molecular systems in which London Dispersion forces are visible using high level quantum mechanical computations.
Results
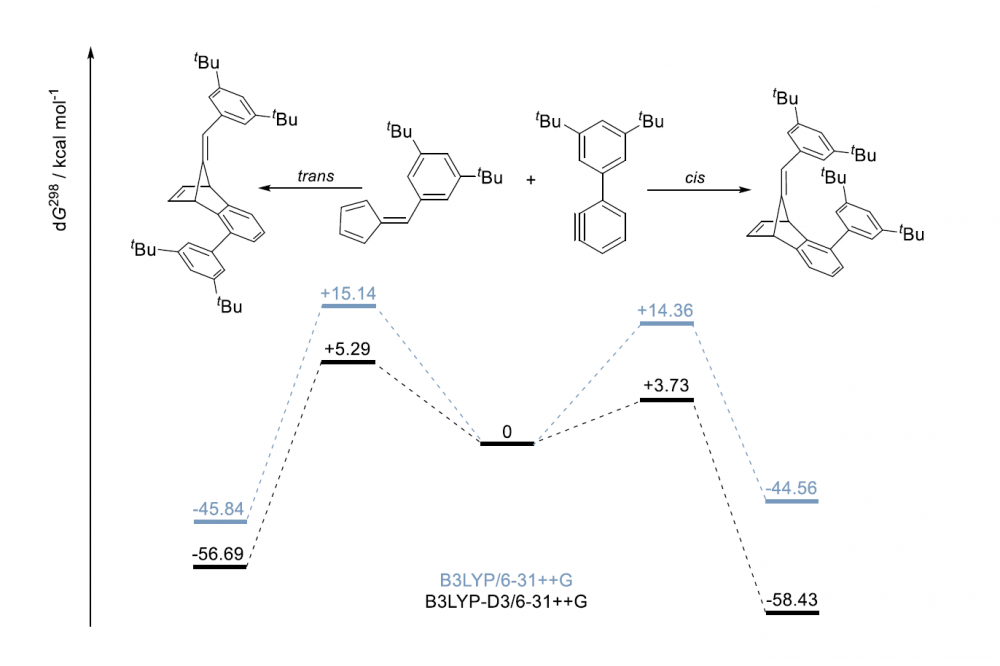
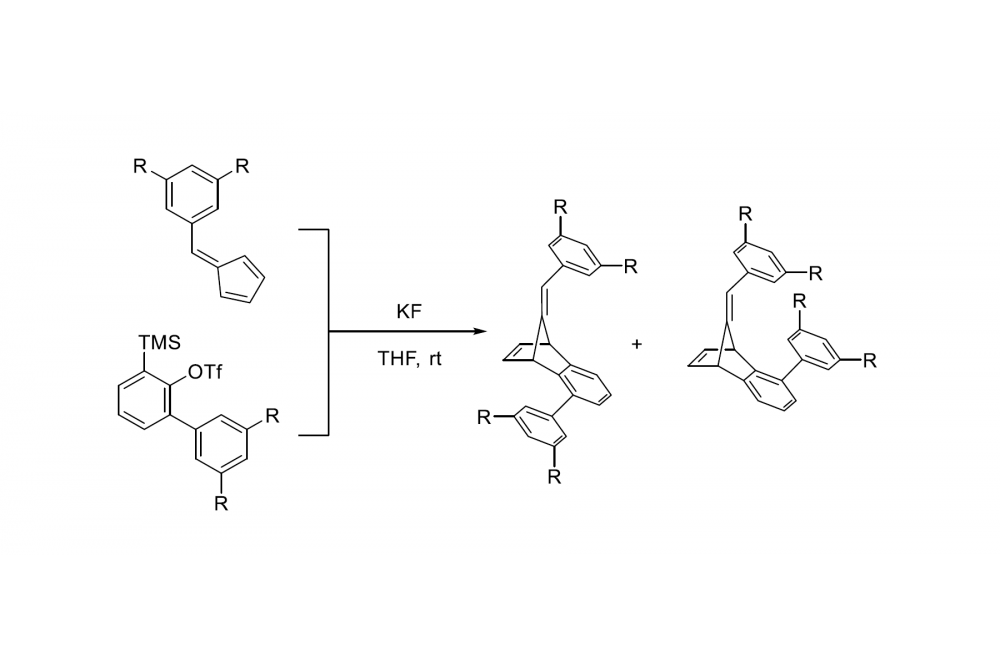
One of the systems we are working on in the Schreiner Group is based on Diels-Alder chemistry. Here, two different products can be formed. The ratio of these products (isomers) highly depends on the transition states of the reaction, whereas most chemists would argue that steric effects dominate and cause selectivity. Our idea here is to stabilize the transition states via London dispersion forces, thereby enriching one isomer. Preliminary computations suggest that, by enlarging the substituents at the molecules, the ratio can be influenced significantly.
Discussion
Using high level computations we can show, that by enlarging our molecules the selectivity of the reaction increases. Additionally, we could demonstrate, that London Dispersion forces the main factor to influence the system. According to the figure below, the cis transition state is lowest in energy, which results in an enrichment of the cis isomer.
Outlook
The knowledge we obtain from this project can be further used to develop catalysts more efficiently. London Dispersion forces may play a significant role in catalyzing reactions of molecules with no functional groups.